Key Insights: Spotlight on BPCRS
Published 2 March 2026
In the latest edition of Key Insights, we are spotlighting BPCRS, looking at:
- their varying types
- their application
- where to find key information and more
Spotlight on BPCRS
When a BP monograph references a BPCRS, it signifies the need for a certified chemical standard to demonstrate compliance with the legally effective monograph. Each BPCRS is a primary standard, meticulously characterised at the ISO 17025-accredited MHRA Laboratory and approved by independent BP experts. These standards are available for purchase and can be incorporated directly into your analytical workflows.

Applications of BPCRS
- Manufacturing and quality control: Supporting quality control testing
- Regulatory testing and inspections: Used by authorities to ensure compliance with pharmaceutical standards
- Research and development in industry and academia: Supporting method development, validation, and scientific research
Types of BPCRS available
The BPCRS catalogue offers a wide range of reference substances to verify the identity, purity, and strength of medicines, along with the performance of analytical methods, including:
- Assay standards: Quantify active substances
- Identity standards: Confirm correct substances
- Mixture standards: Used to assess method performance, system suitability test compliance and/or identify related substances
- Impurity standards: Detect and measure related substances
How to access BPCRS information
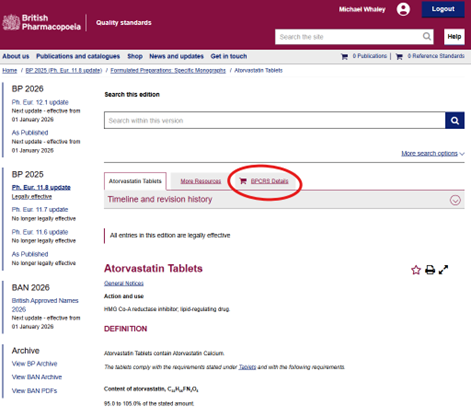
- From the monograph: If you use the BP Online, simply open the BPCRS Details tab within the relevant monograph to view all referenced BPCRS, including catalogue and batch numbers, pack size, CAS number, and quantitative status. Clicking on a specific item reveals further details such as storage/shipping conditions, declared content, and links to the Information Leaflet and Safety Data Sheet.
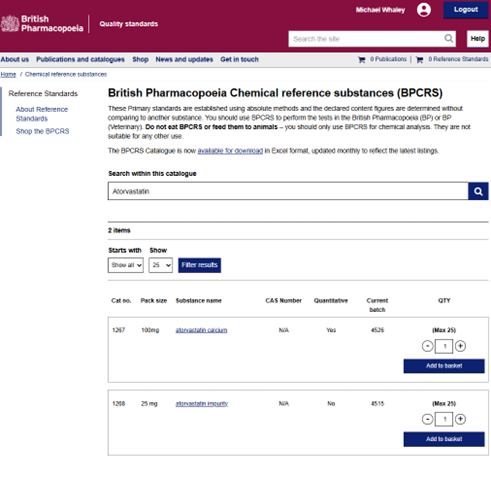
- From the catalogue: The full BPCRS catalogue - comprising over 950 items - can be browsed, searched, and downloaded directly from our website. You can search for BPCRS using the name, the catalogue number, or the Chemical Abstracts Services (CAS) number of the material.
Storage and expiry guidance
All BPCRS are supplied in sealed containers. Long-term storage instructions are provided in the Safety Data Sheet, accessible via the website. Provided the BPCRS remains in its original, unopened packaging and is stored as recommended, it remains valid as long as the batch (lot) number matches the current batch listed online. The BP actively monitors stability and updates batch validity on the website.
Further guidance
For answers to frequently asked questions about storage, expiry, and more, please refer to the “About Reference Standards” section on the BP website or consult Supplementary Chapter III E of the British Pharmacopoeia.
Our BP Scientists Mr Stephen Young and Dr Carolyn Swann, who lead the MHRA’s work on BPCRS, marked World Standards Day in October 2025 by presenting a webinar which can be viewed on the Merck website.
We hope this overview enhances your understanding of BPCRS and encourages you to explore the full range of resources available through the British Pharmacopoeia.

